Nidal Elmoutawakkil1, *, Elhamdi Hanane2, Samira Bellemkhannate3
1Third year resident in the removable prosthodontic department at the D.C.T.C of Casablanca.
2Specialist in removable prosthodontic department at the D.C.T.C of Casablanca.
3Head of the removable prosthodontic department at the D.C.T.C of Casablanca. Director of the national diploma of removable prosthodontic.
*Corresponding Author: Elmoutawakkil N, 3rd year resident in the removable prosthodontic department at the D.C.T.C of Casablanca
Abstract
The coronavirus (COVID-19) appeared in December 2019 in Wuhan China, its mode of spread is mainly through droplets of saliva or nasal secretions when an infected person coughs or sneezes. Its extreme speed of spread across the world added to the current absence of a prophylactic vaccine and effective treatment, prevention remains the best tool against the generalization of such a pandemic. Dental professionals are at greater risk because they work near patients, further exposing them to the risk of contamination from aerosols generated by rotating instruments. Dentists should follow standard precautions to protect themselves against this new virus. This article aims to provide all the safety and preventive measures that can be taken by dental health professionals while providing urgent dental care to patients.
Keywords: coronavirus, FFP2, pandemic, hygiene
Introduction
The Coronavirus belongs to the Coronaviridae family having the RNA genome. According to current reports, the COVID-19 coronavirus affects 210 countries and territories around the world and 2 international means of transport: The Diamond Princess cruiseship hosted in Yokohama, Japan, and Holland America's MS Zaandam cruise ship, with a total number of 6,206,773 confirmed cases (Figure 1) The situation is alarming because of the graph of the average number. of cases, each day is random, especially since the death cases no longer fall within a predetermined age range [1,2].
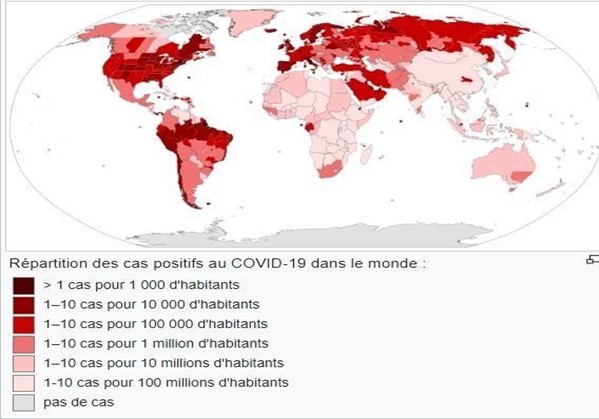
Figure 1: Distribution of positive cases for Covid-19 around the world (source Wikipedia)
Terminology
Initially, the disease was labeled as NCIP (novel pneumonia infected with coronavirus) and the virus was named 2019-n-COV (novel coronavirus 2019) by the World Health Organization. Disease 2019) and the Coronavirus Study Group (CSG) of the International Committee on Virus Taxonomy renamed virus SARS-COV-2 (severe acute respiratory syndrome coronavirus2) [3,4].
Transmission mode
Although the zoonotic origin is now mainly transmitted from humans to humans, it is transmitted by coming into contact with droplets secretion from the upper respiratory tract of infected individuals during sneezing or coughing. One study found that pregnant women infected with SARS-COV-2 showed no evidence of intrauterine transfer10. Another study said there is no clear evidence regarding the optimal time for Childbirth, the safety of vaginal delivery, or whether cesarean delivery prevents transmission [5,6].
Symptomatology
The virus usually presents systemic and/or respiratory manifestations. Some individuals remain asymptomatic and can act as healthy carriers. The most common symptoms are fever, cough, fatigue, sputum, and shortness of breath. Myalgia/arthralgia is less common, as well as migraines, anosmia, and sore throat. Patients may also present bilateral pneumonia, with ground-glass opacity and bilateral uneven shadows being the most common patterns in CT.
Laboratory Results
A study conducted on hospitalized patients demonstrated the following results:
- Lymphopenia
- Increased prothrombin time (PT)
- Increased lactate dehydrogenase
- increased CRP (C-reactive protein) and ESR (erythrocyte sedimentation rate) [8].
Diagnosis:
The definitive test for SARS-COV-2 is the real-time reverse transcriptase-polymerase chain reaction (RT-PCR) test and is believed to have high specificity but low sensitivity, which increases the chances of a false-negative test and therefore more than one test is necessary to confirm the diagnosis [9].
Patient Management Protocol In The Dental Office Since dental professionals are the most affected by this virus, patient management must follow a well-codified protocol with a very specific checklist: Remote screening: An initial screening by telephone is necessary before planning any dental treatment; patients must mention, through a medical questionnaire, their age, medical and travel history, direct contact with a case recognized as positive for Covid-19, any respiratory symptoms (cough, fever, etc.) Any patient complying with the previously mentioned criteria must wait for 2 to 3 weeks to receive any dental treatment.
Patient Assessment
Elective dental care procedures should be postponed in patients with fever (> 100.4 º F = 38 º C) and/or symptoms of respiratory illness for at least 2 to 3 weeks. Each patient should clarify the travel history within the last 14 days and any contact with infected persons.
When confirming appointments or when patients arrive for treatment, appropriate questions should be asked, especially if patients have been in close contact with someone who has been diagnosed or under investigation with COVID-19. A second questioning aims to confirm the urge of the act to limit the risk of cross-contamination; on admission, the extra-body temperature must be collected using a forehead thermometer or with cameras fitted with infrared thermal sensors. Before their admissions to the office, patients must respect hygiene measures (wearing a mask, use of a decontamination mat for the soles on the doormat, manual disinfection by a hydroalcoholic solution) in addition to the patients installed in the room. Waiting must respect the distancing measures (6 feet). Pharmacologic management of infections with antibiotics and pain relievers may relieve symptoms until they are treated other; It will also give dental professionals enough time to reschedule the appointment and screen for emergencies. Patients should use povidone-iodine based mouthwash before starting the procedure. Studies have shown that a pre-operative rinse with a 0.2% povidone-iodine solution mayreduce the coronavirus load in saliva.
On March 17, 2020. “The British Medical Journal” banned the useof ibuprofen in the management of dental pain due to its interference with immune function and recommended acetaminophen as the drug of choice for analgesia in patients infected with COVID-19. The World Health Organization (WHO) approved this recommendation on March 18, 2020 [10].
Patient Management Should Meet the Following Recommendations
- The use of disposable dental equipment is mandatory to contamination: intra-oral x-rays should be avoided as they may cause a gag reflex or cough. Extra-oral radiographs (eg panoramic radiography or cone-beam) should be performed. When intraoral imaging is mandatory, a double barrier is created on the sensors to avoid cross-contamination [11].
- The use of the dam is recommended to minimize the generation of splashing.
Dental procedures that generate higher aerosol content such us ultrasonic instruments, high-speed handpieces, and three- way syringes, should be avoided [12]
Table No. 1: Classification of urgent dental procedures according to the contamination’s risk [13]
|
The operative act |
Niveau de risque |
|
Rapidly spreading infections of the facial spaces / Ludwig's angina / Acute dental cellulitis / Acute trismus. |
Very high |
|
Uncontrolled bleeding from teeth |
Very high |
|
Severe and uncontrolled dental pain, not responding to routine measures. |
High |
|
Trauma involving the face or facial bones. |
Very high |
|
X-rays indicated for facial trauma and in medico-legal situations (panoramic, cone-beam) |
High |
|
Children and adolescents |
|
|
Acute pulpitis |
High |
|
Dental abscess |
Very high |
|
Dento alveolar trauma |
High |
|
Cavities pain requiring temporization |
Very high |
|
Inevitable tooth extractions |
High |
|
Orthodontic procedures (see adults’ section) |
High |
|
Adults and Geriatrics |
|
|
Tooth pain of pulp origin not controlled by counseling, pain relievers, and antibiotics (AAA) |
Very high |
|
Acute dental abscess of pulp origin / periodontal / endo-periodontal / |
Moderate |
|
Vertical dental fracture |
High |
|
Completion of ongoing root canal treatment |
High |
|
Delay of a carious cavity near the pulp but not requiring root canal treatment |
High |
|
Broken prosthetic restoration / fixed prosthesis causing sensitivity of vital teeth / threatening vitality pulpitis / significant difficulty in chewing |
High |
|
Unavoidable dental extractions / post-extraction complications |
High |
|
Dental abutments/implants already prepared to receive crowns |
Very high |
|
Peri-implant infections endangering stability |
High |
|
Pericoronitis / operculetomy |
High |
|
Oral mucosal lesions requiring a biopsy |
High/moderate |
|
Long-standing jaw cysts and tumors with abrupt changes |
High |
|
Sharp teeth / trigeminal neuralgia |
Moderate |
|
Orthodontic devices piercing or injuring the oral mucosa |
Moderate |
|
Moderate orthodontic treatment with iatrogenic effects |
Moderate |
|
Placement of clear aligners |
Moderate |
|
Patients with skeletal anchors |
Moderate |
|
Repair of broken complete dentures |
High |
|
Problems related to implant-supported prosthesis |
High |
|
Infections of the oral mucous membrane such as candida |
High |
|
Oral mucosal lesions showing sudden changes or suspected malignancy requiring biopsy |
High |
|
Patients under medical treatment |
|
|
Diabetic patients requiring treatment for periodontal disease |
High |
|
Dental treatment for patients who are candidates for heart surgery |
Very high |
|
Hospitalized patients requiring dental treatment |
Very high |
|
Radiotherapy/organ transplant candidate patients requiring dental treatment |
Very high |
Preparation of the Premises: Recommendations of The High Society REHVA "The Federation Of European Heating, Ventilation And Air Conditioning Associations"
The management of ventilation and air quality in independent dental clinics must comply with the following recommendations:
- Maintain natural air circulation by frequently opening windows and using an independent exhaust fan to extract room air into the atmosphere.
- Avoid the use of a ceiling fan when performing the procedure.
- Place a table ventilator behind the operator and allow air to circulate to the patient. A powerful exhaust fan should be located to create a one-way airflow away from the patient.
- The air conditioning system must be frequently maintained, and the filters cleaned.
- The use of a portable indoor air purification system equipped with a HEPA filter and UV light is recommended; In central AC buildings, the recirculation system must ensure the blocking of return vents air in the patient area by allowing fresh air to enter the rooms through half-opened windowsand doors.
- It is recommended that any ventilation or air conditioning system operating normally with a recirculation mode is now configured to operate in the open air when possible. In buildings with mechanical ventilation systems, extended run times are recommended. In demand control systems, CO2 setpoints should be fixed at 400 ppm to increase the outdoor airflow. Ventilation should be maintained for longer, with lower ventilation rates when people are away. It is not recommended to turn off ventilation systems in buildings, even those temporarily vacated, but to run them continuously at reduced speeds. Recirculation of air between spaces, rooms, or areas occupied by different people should be avoided. However, in the case of any system serving only one space, the partial recirculation of the air inside it, for example by a local fan coil, is of less concern. The reason for this is that the main objective is to maximize the rate of air exchange with the outside air and to minimize the risk of stagnant air pockets.
- Where planned cleaning or maintenance of ventilation systems is required, it should be undertaken following agreed industry guidelines, including those relating to siteoperations under social distancing requirements. Appropriate personal protection equipment (PPE) should be worn and all materials, including old filters, should be carefully packaged, and disposed of safely.
- In poorly ventilated spaces with high occupancy and where it is difficult to increase ventilation rates, it may be appropriate to consider the use of air cleaning and disinfection devices. The most suitable devices are probably local HEPA filtration units or those using UV germicidal radiation (GUV). GUV devices use radiation in the UV-C spectrum and have been shown to inactivate coronaviruses, although there is no specific evidence yet for the effectiveness of UV-C irradiation on SARS-COV-2. There are currently uncertainties about a variety of factors affecting UV performance, including dosage, wavelength, and exposure time. Besides, consideration should be given to the specific configuration of the room and system, airflow, distribution, and humidity. Proper safety interlocks must be installed to ensure that UV cleaning equipment is not used when people may be exposed to radiation. They can be applied as an upper chambersystem or as a stand-alone consumer unit, but these must be sized correctly for the room, as many do not have the flow to be effective in larger spaces. UV-C in ducts is not recommended for controlling disease transmission except for decontaminating the recirculated air. Devices emitting ozone or other potentially hazardous by-products should not be used in occupied spaces.
- Working spaces must be ventilated with outside air, the ventilation must be set at a nominal speed at least 2 hours before using the building to shift to a lower speed 2 hours after that.
- At night and on weekends the ventilation must not be deactivated the ventilation systems must be functional but at a lower speed [14].
Maintenance of The Reception and Waiting Room
- Post visual alerts at the entrance to the facility and in strategic areas (for example, waiting rooms or elevators) regarding respiratory hygiene, cough etiquette, social distancing, and disposal of contaminated items in the trash.
- Install a glass or plexiglass barrier at the reception, preferably with a bidirectional speaker system.
- Make sufficient tri-layer masks, disinfectants, and toilet paper available at the registration desk, as well as at nearby hand hygiene stations.
- In the waiting room, move the chairs a meter apart.
- All areas should be free of magazines, toys, TV remotes, or similar items.
- Contactless payment methods are preferred.
- A dustbin with a lid should be available at the triage where patients can throw away used tissues.
- Patients should minimize or eliminate wearing jewelry and carrying extra accessory bags, they should also wear a face mask throughout the transport to the treatment room, their body temperature should be checked at the clinic. Home.
- The use of the toilet in the dental office should be prohibited for patients.
- The assistant should offer patients a 0.5% polyvidone iodine rinse solution distributed in the oral cavity for 30 seconds, then as a gargle in the back of the throat for 30 seconds.
- Consent and patient declaration must be obtained on paperor electronically [15].
A Zone Dedicated To Putting On And Taking Off Personal Protective Equipment (PPE) Changing rooms should be available so that all working staff can wear surgical pajamas, surgical gown, and clinic shoes, this room should be divided into two areas: one for dressing and another for removing PPE.
The Area Dedicated to Sterilization
A dedicated and trained person should be available to undertake transportation, cleaning, drying, packaging, sterilization, storage, and sterilization quality testing according to standard guidelines and manufacturer's instructions. Sufficient and dedicated space for the storage of additional PPE items and sterilization and disinfection instruments.
Equipment and Instrumentation
opt for taps with elbow handles or fitted with sensors Favor paper towels.
Install fumigation systems.
Addition of a secondary high flow oral suction. Implementation of an indoor air purification system.
The dental chair's water lines should be fitted with retraction valves. Handpieces must be fitted with anti-retraction valves.
chemicals required for disinfection and decontamination of PPE.
Patient Management Protocol:
1. Hand washing: for 40-60 seconds
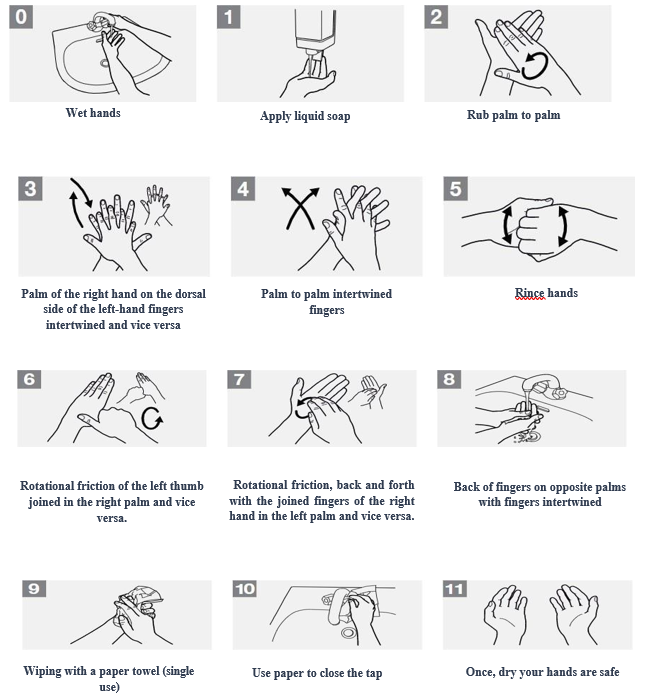
Figure No. 2: handwashing protocol
2. Disinfection of dry hands using a hydroalcoholic solution for 20 to 30s:
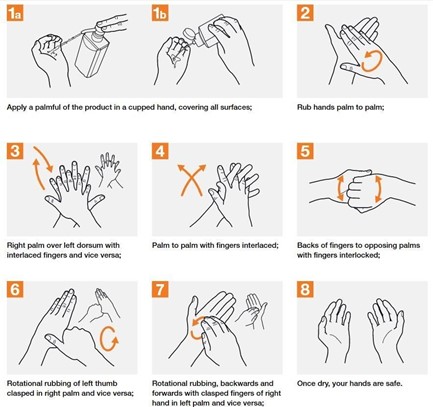
Figure No. 3: hand disinfection protocol [13]
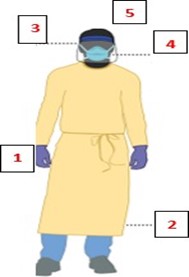
3. Putting on personal protective equipment:
After hands washing and hydroalcoholic decontamination, the dressing sequence is as follows:
- Identify and assemble the appropriate PPE to put on, making sure you choose the right size for the gown and attach all the ties on their outer face.
- Put on the cap making sure it covers all of the hair.
- Put on an N95 / FFP2 filtering respiratory mask. If you have a respirator with a nasal nozzle, it must be fitted to the nose with both hands, not folded or putt in a tent pole; The respirator/face mask should be extended under the chin, mouth, and nose should be protected (Figure 3).
- Perform a user tightness check each time the respirator is put on; the mask fasteners should be attached to the upper part of the skull and the base of the neck.
- Put on protective goggles or a face shield, making sure the respirator does not interfere with proper positioning of the eye protection and that the eye protection does not affect the fit or seal of the respirator. Face shields provide full-face coverage without fog.
- Put on clean gloves covering the wrist of the gown [13,16].
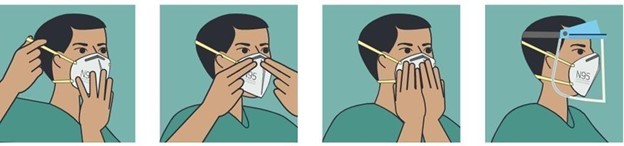
Figure No. 4: Correct placement of the FFP2 mask [13]
4. Removal of PPE:
- Remove gloves, making sure they are tight to avoid further manual contamination.
- Remove the gown by loosening all ties as soon as you reach the shoulders and pull gently down and away from the body. Rolling it up is an acceptable approach. Throw in the trash.
- Remove the visor or goggles without touching the front by grasping the strap and pulling up and away from the head.
- Remove and discard the mask as follows: remove the lower strap by touching only the strap and carefully bring it over the head then grasp
- the upper strap and gently bring it over the head, finally remove the mask of the face without touchingits front.
- Remove the cap and throw it in a trash can.
- Perform manual washing with soap and water [13].
Protocol for Decontamination and Disinfection of The Operating Room After the Patient's Departure
- Once the patient has left the operating room, the manual instruments are immediately collected and rinsed with running water to remove all organic debris, then the standard sterilization protocol is initiated.
- All rotary instruments should be irrigated and flushed with a disinfectant solution for 30 to 40 seconds; their external surface should also be decontaminated before they are autoclaved.
- Remove and discard all disposable consumables, such us: suction cannulas, paper drapes, and plastic cups, then irrigate the internal suction pipes with a 2% sodium hypochlorite solution for a period of 30 to 40 seconds, the latter is also used to wash the spittoon (even the use of the latter is prohibited by the patient).
- The dental chair, as well as all chairs, stools, and mobile units within a perimeter of 3 meters, must be decontaminated by a disinfectant solution and disposable paper going from the internal surfaces to the external ones without forgetting the armrests, headrests, whips, body of the air and water syringes and all the vertical surfaces surrounding the working area.
Decontaminate the Floor in Two Steps
- First, with a 1% sodium hypochlorite solution with a contact time of 10 minutes, thwart using the same piece of tissue in the other rooms of the dental office.
- We should start with the farthest corner of the room and work towards the door; This procedure should be repeated after each patient, any major splash, or every two hours.
- The rest of the surfaces are decontaminated with freshly prepared 1% sodium hypochlorite (Contact time: 10 minutes).
- Wet dusting should be performed in straight, overlapping lines before and after each patient.
- Delicate electronic equipment should be wiped down with a 60-90% alcohol solution before each contact with the patient.
- The survival time of the Covid19 virus can be soar to 9 days at room temperature, with a greater preference for humid environments, thus dry conditions must be maintained to prevent the spread of this pandemic [17].
Decontamination of Impressions
- The decontamination method will vary depending on the impression material used except in the first step which consists of rinsing it under running water, which has to be done immediately after removal from the oral cavity, under running water until all traces of blood, saliva, and other organic matters have disappeared. This simple rinse is considered the most important step of the decontamination process, as it reduces contamination by 90% in 15 seconds
- Rinsing helps remove organic blood and salivary debris, phlegm, and bacterial plaque. Organic waste may indeed reduce the action of disinfectants, such as sodium hypochlorite, phenols, iodophors products, or alcohol see their actions annihilated by the presence of organic blood or salivary residues.
Before proceeding to the next step, excess water should be removed by vigorously shaking the impression. The reason is that excess water could reduce the effectiveness of the disinfectant solution by diluting it. It is important to remember that following the concepts of individual protection in terms of hygiene, this step must be carried out by personnel wearing gloves as well as protective glasses or a visor mask [18].
Alginates
They are the most studied materials given their greater susceptibility to contamination and disinfection procedures. Indeed, alginate is the material which retains the most microorganisms and which is the most subject to deformation; This is explained by its permeability linked to its hygroscopic components. Great importance should therefore be attached to the possible consequences of disinfection procedures [19].
Decontamination is carried out either by:
- Spraying of a solution based on 2% glutaraldehydes, sodium hypochlorite at concentrations varying from 0.5 to 5% or with mixtures of alcohol and aldehyde or iodine derivatives,
- Immersion less than 10 minutes in these solutions,
- By comparing the advantages and inconveniences of these two methods, the technique that seems best suited to this type of impression (intermediate method) consists of:
- Cleaning the impression under running water then shake it to remove excess water.
- Immersing the impression for a few seconds in a 0.5% sodium hypochlorite solution.
- Wrapping with gauze or absorbent paper the impression which will be sprayed with a spray soaked in the same solution.
- Placing the treated impression in a hermetically sealed plastic bag for 30 minutes.
- Then rinse it under cold water to remove all the disinfectant solution.
- Shake it.
- Place the impression in a hermetically sealed sterile bag.
- Put everything in a box and send it to the laboratory.
Silicones:
All the silicone families are made up of hydrophobic impression materials. Therefore, they retain fewer microorganisms. Their decontamination is simpler and above all much less controversial compared to hydrocolloids.
The method of decontaminating silicone impressions should go through the following steps:
- Rinse the impression under running water for at least 15 seconds.
- Lightly dry the impression to remove excess water.
- Immerse the impression in a closed tank for 30 minutes in a solution of 2% glutaraldehyde or 0.5% sodium hypochlorite.
- Rinse the impression again under running water.
- Dry the impression.
- Place the impression in a hermetically sealed sterile bag.
- Put everything in a box and send it to the laboratory.
For high-risk patients, the immersion should be prolonged up to 2 hours. Therefore, when there is a known risk, only silicone impressions should be recommended [20].
Polysulfides:
According to HELFER (2010), polysulfides will undergo the same treatment as silicones. These hydrophobic materials retain fewer microorganisms. The impressions will, after rinsing, be immersedfor 30 minutes (0.5% sodium hypochlorite or 2% glutaraldehyde). They will finally be rinsed and dried [21].
In the study by HATRICK and EAKLE (2015), it is stated that polysulfides should be decontaminated by immersion for 10 to 30 minutes in 10 volume sodium hypochlorite or phenolic complexes [22].
Polyethers:
The polyether impression must therefore be decontaminated by spraying with a solution based on 2% glutaraldehyde or sodium hypochlorite at 0.5%. The intermediate method proposed for alginates can perfectly be used for this type of impression material [23]. Hatrick and Eakle (2015) recommend the following disinfection methods for polyethers: immersion less than 10 minutes in 10 volume sodium hypochlorite (1:10) or spraying with sodium hypochlorite at 10 volumes (1:10) [22]. According to Laviole (2015), polyethers should be disinfected after rinsing with running water by spraying with 2% glutaraldehyde [24].
Zinc Oxide Eugenol:
This material does not undergo any modification with glutaraldehyde, while sodium hypochlorite should be avoided. The impression must therefore be decontaminated by immersion in a 2% glutaraldehyde- based solution for 10 to 60 minutes [21]. The following table shows the different specificities of the decontamination according to the material used for impressions in the prosthodontics.
Table No. 2: Summary of techniques and products used for the decontamination of impressions in prosthesis [25].
|
Material |
Methods |
Used products |
|
Irreversible hydrocolloids: alginate |
60mn spray foriodine derivatives,10 to 30 for others.Immersion:afew secondsin a hypochlorite solution 15minutes for other solutions |
2% glutaraldehyde, 2.5% 2%, 1%, 5.25% hypochlorite A mixture of alcohol and aldehyde Iodine derivatives |
|
Condensation curing silicone |
Immersion 15 mn 60 mn |
Aldehyde-based product 2% glutaraldehydes Hypochlorite (10,000 ppm chlorine) Iodine derivatives |
|
Addition curing silicone |
Immersion 10mn 15 mn 60 mn |
|
|
Polysulfones |
Immersion 10 to 60 minutes |
2% glutaraldehyde Hypochlorite (10000 ppm chlorine) Iodine derivatives |
|
Polyethers |
Spray Immersion for a few seconds and spraying hypochlorite |
2% glutaraldehyde solution 1% sodium hypochlorite |
|
Eugenol zinc oxide |
Immersion 10 to 60 minutes |
Glutaraldehydes 2% Iodine derivatives |
Even if the impression processing has been done correctly, the risks of cross-contamination are not eliminated; This is the reason why working models must also undergo a decontamination treatment during and after their development.
It is proven that some of these microorganisms are also found on the plaster models cast from the impressions concerned. Also, some of these microorganisms may persist on plaster models up to seven days after casting. Cast and models may contain opportunistic or pathogenic microorganisms that can be disseminated in the laboratory and cause cross-contamination patient-personal or patient-patient. This contamination occurs if the instruments used in the laboratory are not disinfected between the concerned patients, as well as the working surfaces.
It is also important to avoid the risk of chemical interaction between decontamination products used in the dental office and the laboratory; for example, contact of hypochlorite with other chemicals may reduce its disinfectant effectiveness. Besides, if chlorhexidine comes in contact with minerals, hard water and if the pH is above 8, it causes precipitation of chlorhexidine, biguanides are incompatible with halogens, aldehydes, mercurials, anionic and nonionic surfactants, soaps, low-density polyethylene containers; Finally, the incompatibilities of phenolic derivatives and phenols have been found with: iron salts, hypochlorites, alcohols (inhibitions), and ammoniums quaternary.
A dental impression cannot therefore randomly undergo two successive disinfectant treatments, confirming the need for good communication between the laboratory and the practitioners to prevent any deteriorations [23].
Decontamination in The Laboratory Will Take Place:
- During the cast:
Incorporation of a decontaminating solution into the liquid/powder mixture.
- After preparation:
The Decontamination of Plaster Models Can Be Done Either By:
- Spraying with a 0.5% sodium hypochlorite solution.
- Immersing the models for 10 minutes in the same solution.
- Using a solution of saturated calcium sulfate and sodium hypochlorite at 5.25% [26].
The Plaster
Disinfection of plaster used as impression material is delicate. It is possible to add a disinfectant solution to the casting plaster. Immersion of a plaster impression in sodium hypochlorite for 10 minutes is not recommended as damage may occur [21].
Individual impression trays
They are decontaminated by rinsing under running water followed by immersion or spraying with a solution of hypochlorite or phenol [22].
Thermoplastic materials (waxes)
As with impressions, waxes rims should be rinsed with running water before being decontaminated to remove saliva and any organic debris. Since they cannot undergo heat sterilization, disinfectant solutions are used. However, alcohol-based substances should be avoided because alcohol is a solvent for waxes; its use would reduce the accuracy of the rim’s recordings.
The protocol is as follows: rinse-spray-rinse-spray.
The material is then enclosed in a sealed plastic bag for the time recommended by the manufacturer.
According to HELFER (2010), waxes should be disinfected using chlorhexidine (concentration not specified). They will be rinsed with water, immersed in a solution of chlorhexidine, and then rinsedagain carefully before being dried. In the dental department of the university hospital center in Nancy, the waxes are decontaminated in Amukine® (sodium hypochlorite) for 3 minutes [21].
Full dentures
Heat sterilization of resins is impossible because the material undergoes much greater distortions. The decontamination technique is as follows:
- Rinsing with running water,
- Immersion in 5.25% sodium hypochlorite or iodophors for 5 minutes,
- Rinsing with running water,
- Ultrasonic cleaning in distilled water,
- Drying of the dentures which are then sealed in a sterile bag,
- Put everything in a box [24].
Resin partial dentures
The decontamination technique is identical to the one used for full dentures, but corrosion of the metal parts through the hypochlorite might occur.
Metallic partial dentures
The metal frame must follow the chain of sterilization in the autoclave or cold disinfection by immersion in 70 % alcohol.
The entire metallic partial denture can be decontaminated by immersion in a solution based on iodine derivatives [27].
Ceramic and metallic crowns:
They must follow the chain of sterilization by autoclave or cold disinfection following immersion in 70% alcohol [21].
Conclusion:
The worldwide spread of the Covid-19 virus has affected the global economy in every way. Until this pandemic cloud is brought under control, dentists should defer elective treatment. Emergency procedures should be performed following universal precautions and appropriate patient screening: some of these are asymptomatic carriers since the incubation period can range from 0 to 24 days with mild symptomatology. Thus, all dentists must protect themselves and their patients and working staff while respecting hygiene’s universal recommendations.
References
- Versaci MB (2020) “ADA releases coronavirus handout for dentists based on CDC guidelines”.
- Michael Day (2020) “Covid-19: ibuprofen should not be used for managing symptoms, say doctors and scientists”:1.
- Zhu N, Zhang D, Wang W, Li X, Yang B, et al. (2020) A Novel Coronavirus from Patients with Pneumonia in China, 2019. N Engl J Med 382(8): 727-733.
- Gorbalenya AE, Baker SC, Baric RS, de Groot RJ, Drosten C (2020) Severe acute respiratory syndrome-related coronavirus: The species and its viruses, a statement of the Coronavirus Study Group 32(4): 1-9.
- Heymann DL, Shindo N (2020) COVID-19: What Is Next for Public Health? 395(10224): 542-545.
- Chen D, Yang H, Cao Y, Cheng W, Duan T, et al. (2020) Expert consensus for managing pregnant women and neonates born to mothers with suspected or confirmed novel coronavirus (COVID-19) infection. Int J Gynaecol Obstet 149(2):130-136.
- Wang D, Hu B, Hu C, Zhu F, Liu X,et al. (2020) Clinical Characteristics of 138 Hospitalized Patients With 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, China. JAMA 323(11): 1061-1069.
- Rodrigues JCL, Hare S, Edey A, Devaraj A, Jacob J et al. (2020) An update on COVID-19 for the radiologist A British society of Thoracic Imaging statement 75(5): 323-325.
- Kanne JP, Little BP, Chung JH, Elicker BM, Ketai LH (2020) Essentials for Radiologists on COVID-19: An Update- Radiology Scientific Expert Panel. Radiology 296(2): 113- 114.
- Peng X, Xu X, Li Y, Cheng L, Zhou X et al. (2020) Transmission routes of 2019-nCoV and controls in dental practice. Int J Oral Sci. 12(1): 9.
- Hokett SD, Honey JR, Ruiz F, Baisden MK, Hoen MM (2000) “Assessing the effectiveness of direct digital radiography barrier sheaths and finger cots.” J Am Dent Assoc 131(4): 463-467.
- Dr. Sharma Y, Dr. Marwaha J, Dr. Bishnoi RR, Goyal R, Dr. Biswas A (2019) Imaging in Latest Dentistry-A Review. Int J Inn Res Med Sci. 4(12): 712-717.
- Guidelines for Dental Professionals in Covid-19 pandemic situation, department of health research Ministry of Health and Family Welfare, Government of India, May 2020.
- REHVA COVID-19 guidance document, April 3, 2020.
- Fallahi H.R, Okeyhan S, Zandian D, Kim S.G, Cheshmi B (2020) Being a front-line dentist during the Covid-19 pandemic: a literature review Maxillofac Plast Reconstr Surg. 42(1): 12.
- Versaci MB (2020) ADA releases coronavirus handout for dentists based on CDC guidelines: American Dental Association.
- Barabari B, Moharamzadeh K (2020) Novel Coronavirus (COVID-19) and Dentistry-A Comprehensive Review of Literature Dent J 8(2): 53.
- Helfer M, Fabbro SD (2015) Asepsis in prosthesis in the dental office Profession Dental Assistant: Nov-Dec. 12-16.
- Suprono MS, Kattadiyil MT, Goodacre CT, Winer MS (2020) Effect of disinfection on irreversible hydrocolloid and alternative impression materials and the resultant gypsum casts. J Prosthet Dent. 108(4): 250–258.
- Walker MP, Rondeau M, Petrie C, Tasca A, Williams K (2007) Surface quality and long-term dimensional stability of current elastomeric impression materials after disinfection. J Prosthodont. Sep-Oct 16(5): 343-351.
- Helfer M. Respect for asepsis in the prosthetic development chain. JPIO. 2010 ; Special issue Hygiene and asepsis (HS): 41-50.
- Hatrick CD, Eakle WS (2015) Dental Materials: Clinical Applications for Dental Assistants and Dental Hygienists. Third edition. Elsevie Health Sci.
- Yilmaz H, Aydin C, Gul B, Yilmaz C, Semiz M (2007) Effect of disinfection on the dimensional stability of polyether impression materials. J Prosthodont. Nov-Dec. 16(6): 473- 479.
- Laviole O. Creation of working models in joint prosthesis. EMC Oral Medicine. 2015 ; 10(4) : 1-11.
- Muller M, Bolla M (1995) Decontamination of impressions. Aboutostomatol, 189 : 51-68.
- Missika P, Drouhet G (2001) Hygiene, asepsis, ergonomics. A permanent challenge, JPIO, Edition CdP 120.
- Hue O, Berteretche MV (2003) The laboratory Complete prosthesis, Quintessence international, PP: 263-280.



