Chun Peng Goh1, *, Samantha Wei Lee Yong2, Su Lone Lim2, Ting Ting Feng2, Ira Siyang Sun2, Shiong Wen Low2
1Division of Neurosurgery, Department of General Surgery, National University Hospital, Singapore.
2Division of Neurosurgery, Department of General Surgery, Ng Teng Fong General Hospital, Singapore.
*Corresponding Author: Chun Peng Goh, Division of Neurosurgery, Department of General Surgery, National University Hospital, Singapore.
Abstract
Background
Gamma knife radiosurgery (GKS) has increasingly been utilized as primary treatment for a Dural-based lesion especially when lesions are in challenging locations.
Case description
We present a case of a patient with a parasagittal Dural-based lesion who was initially treated with gamma knife radiosurgery in 2016 as presumed meningioma. The lesion was located over the left precentral gyrus and had responded well to GKS with radiological proof of shrinkage of size. However, it evolved after 3 years after GKS and the rate of progression since was rapid and finally, she underwent surgical resection. Histology confirmed anaplastic hemangiopericytoma (WHO Grade III).
Conclusion
This case report highlights the pitfall of GKS for which treatment was provided based on the assumption that the lesion is a benign meningioma. As such, patients should be counselled appropriately prior to receiving GKS treatment as a primary treatment for a Dural-based lesion. We propose a long-term follow-up even after radiological proof of response of the tumour to GKS.
Keywords: Dural-based lesion, gamma knife radiosurgery, resection
Introduction
Over the last two decades, intracranial Dural-based lesions are increasingly being treated with gamma knife radiosurgery (GKS). Even though surgical resection is the mainstay of treatment, it carries inherent risks especially when lesions are in challenging locations. GKS has even been proposed as the treatment of choice, supported by evidence of its effectiveness and safety profile. We would like to highlight a pitfall of GKS as the primary treatment of Dural-based lesions with this case report.
Case report
We present a case of a 63-year-old female who first presented in May 2016 with generalised tonic-clinic seizures which spontaneously aborted after one minute. This was preceded by several months of recurrent focal seizures with retained awareness described as right lower limb stiffness. There were no focal neurological deficits in between episodes.
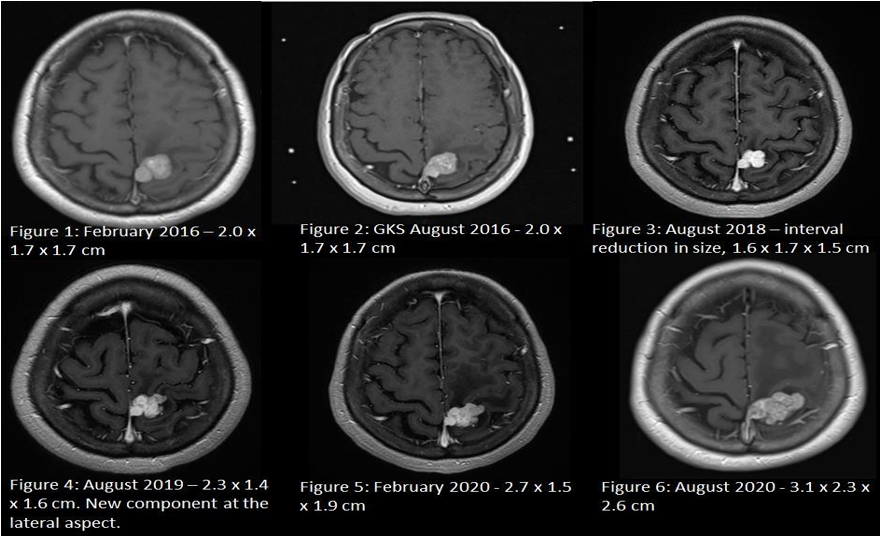
A magnetic resonance imaging (MRI) of the brain revealed a 2.0 x 1.7 x 1.7 cm left posterior parasagittal Dural-based contrast-enhancing lesion with perilesional oedema (Figure 1) which was presumed to be a benign meningioma. Her seizures were managed with levetiracetam and sodium valproate.
Surgical resection was offered as the treatment of choice. However, she was not keen on surgery due to the inherent risks of surgical resection, especially that the tumour is located near the pre-central gyrus. She had opted for gamma knife radiosurgery (GKS) as the alternative treatment and received a radiation dose of 13 Gy to the tumour in August 2016 (Figure 2).
Subsequent interval scans up till August 2018 showed a reduction in the size of the tumour to 1.7 x 1.6 X 1.4 cm (Figure 3). However, between these periods, she had recurrent breakthrough focal seizures with similar semiology contributed by non-compliance of anti-epileptic medications.
A repeat MRI Brain in August 2019 showed that the tumour had unfortunately grown to 2.3 x 1.4 x 1.6 cm (Figure 4), with a new enhancing component at the left lateral aspect. Another MRI Brain in February 2020 demonstrated progression to 2.7 x 1.5 x 1.9 cm (Figure 5) with increased perilesional oedema.
She had also experienced more frequent focal seizures and developed right ankle weakness. She was offered repeat GKS and surgical resection and had opted for surgical resection in August 2020.
Volumetric MRI brain done the day prior to surgery showed further increase in the size of the tumour to 3.1 x 2.3 x 2.6 cm (Figure 6). She underwent craniotomy and gross total resection of the tumour was achieved. Histology confirmed the diagnosis of an anaplastic solitary fibrous tumour or hemangiopericytoma (WHO Grade III) which was positive for CD34 and STAT6 expression and PR negative.
Post-operatively, she recovered well with minimal reduction in right lower limb strength. A computed tomography scan of the thorax, abdomen and pelvis was done which did not show any extra-cranial involvement. She is planned for adjuvant radiotherapy to the surgical cavity.
Discussion
The predominant treatment of intracranial meningioma is surgical resection. The aim of surgery is to achieve maximal resection of the tumour and to maintain intact neurological function. As surgery carries inherent risks, gamma knife radiosurgery (GKS) is increasingly being considered a treatment option. GKS has been applied frequently in patients in whom the meningioma is in a challenging location and for small solitary tumours as well as adjuvant therapy after incomplete resections. [5]
Effectiveness of GKS for presumed intracranial meningioma
Many series have suggested that GKS is effective for controlling tumour growth. Pollock et al’s experience [9] of GKS on 251 presumed intracranial meningiomas showed that the 10-year local control rate was 99.4 %. In another retrospective study of 628 samples [6], the overall tumour control rate was 95 % with a median follow up of 37 months. Jang et al suggested that GKS for intracranial meningioma is safe and effective with successful long-term outcomes. A recent retrospective study [3] of a series of 130 patients suggested that GKS can be considered as the primary or adjuvant management of intracranial benign meningioma if tumour volume is less than 10cm3, pre-GKS KPS of more than 90 and no pre-GKS cranial nerves deficit.
GKS as treatment of choice
Importantly, Kondziolka et al [7] found no difference in long-term (10 years) tumour control rates between patients who had undergone craniotomy before radiosurgery (89 %) and patients who underwent primary radiosurgery (93.1 %).
Pollock et al [10] conducted a comparison between GKS and surgical resection for a benign or presumed benign meningioma. It showed that progression-free survival rate after GKS was equivalent to that after a Simpson Grade 1 resection of the tumour and was superior to Grade 2 and 3-4 resections. GKS was recommended as the primary treatment modality for meningiomas where Simpson grade 1 resection was not achievable.
Low potential risk of malignant transformation
A retrospective study by Wolf et al [12] showed that the overall incidence of radiosurgery-associated malignancy was 6.80 per 100 000 patients-years or a cumulative incidence of 0·045 % over 10 years. It is concluded that in patients treated with GKS, the estimated risk of an intracranial secondary malignancy or malignant transformation of a benign tumour remains low at long-term follow-up.
Another retrospective cohort study [11] Based on a database with 5000 patients and 30,000 patient-years of follow-up indicated no increased risk of malignancy, providing evidence for the safety of GKS.
Hemangiopericytoma (HPC) accounts for less than 1 % of intracranial tumours and approximately 2.5 % of all meningeal tumours. [4] Radiologically, it is difficult to be distinguished from a meningioma. [5] The highly vascular nature of HPCs results in their being avidly enhancing and well-demarcated on MRI. These tumours tend to be very sensitive to radiosurgery, often exhibiting rapid and dramatic regression. [2,11]
In our case, treatment for this patient’s dural-based lesion was indicated as she was symptomatic. As the tumour was near the motor strip, GKS was the acceptable alternative to surgical resection. The tumour responded well to GKS with radiological proof of shrinkage of size. However, the tumour evolved after 3 years after GKS and the rate of progression since was rapid. One important observation was that there was minimal bleeding during piecemeal resection of the tumour which is likely related to the effects of GKS.
Conclusion
This case report highlights the pitfall of GKS for which treatment was provided based on the assumption that the lesion is a benign meningioma. As such, patients should be counselled appropriately prior to receiving GKS treatment as a primary treatment for a Dural-based lesion. We propose a long term follow up even after radiological proof of response of the tumour to GKS.
Conflicts of interest/disclosure
The authors declare that they have no financial or other conflicts of interest in relation to this research and its publication.
References
- Akiyama M, Sakai H, Onoue H, Miyazaki Y, Abe T (2004) Imaging intracranial haemangiopericytomas: study of seven cases. Neuroradiology. 46(03): 194–197.
- Coffey R, Cascino T, Shaw E (1993) Radiosurgical treatment of recurrent hemangiopericytomas of the meninges: preliminary results. J Neurosurg 78(6): 903-908.
- Ge Y, Liu D, Zhang Z, Li Y, Lin Y, et al. (2019) Gamma Knife radiosurgery for intracranial benign meningiomas: follow-up outcome in 130 patients. Neurosurg Focus. 46(6): E7.
- Ghose A, Guha G, Kundu R, Tew J, Chaudhary R (2017) CNS hemangiopericytoma: a systematic review of 523 patients. Am J Clin Oncol 40(3): 223–227.
- Iwai Y, Yamanaka K, Ikeda H (2008) Gamma Knife radiosurgery for skull base meningioma: long-term results of low-dose treatment. J Neurosurg. 109(5): 804-10.
- Jang CK, Jung HH, Chang JH, Chang JW, Park YG, et al. (2015) Long-Term Results of Gamma Knife Radiosurgery for Intracranial Meningioma. Brain tumor Res Treat. 3(2): 103-7.
- Kondziolka D, Patel AD, Kano H, Flickinger JC, Lunsford LD (2016) Long-term outcomes after gamma knife radiosurgery for meningiomas. Am J Clin Oncol. 39(5): 453-7.
- Payne BR, Prasad D, Steiner M, Steiner L (2000) Gamma surgery for hemangiopericytomas. Acta Neurochir (Wien). 142(5): 527-536.
- Pollock BE, Stafford SL, Link MJ, Garces YI, Foote RL (2012) Single-fraction radiosurgery for presumed intracranial meningiomas: efficacy and complications from a 22-year experience. Int J Radiat Oncol Biol Phys. 83(5): 1414-8.
- Pollock BE, Stafford SL, Utter A, Giannini C, Schreiner SA (2003) Stereotactic radiosurgery provides equivalent tumor control to Simpson grade 1 resection for patients with small-to-medium-size meningiomas. Int J Radiat Oncol Biol Phs. 55(4): 1000-5.
- Rowe J, Grainger A, Walton L, Silcocks P, Radatz M, et al. (2007) Risk of malignancy after gamma knife stereotactic radiosurgery. Neurosurgery. 60(1): 60-5
- Wolf A, Naylor K, Tam M, Habibi A, Novotny J, et al. (2019) Risk of radiation-associated intracranial malignancy after stereotactic radiosurgery: a retrospective, multicentre, cohort study. The Lancet Oncology. 20(1): 159-164.