Adrian Hoenle, MD BSc1, *, Florian Kister MD2, Stephan Lorenz MD3, Claus-Martin Muth MD PhD4, Robert Schappacher MD5
1Division of ENT/Head and Neck Surgery, Marienhospital Stuttgart, Germany
2Division of Oral and Maxillofacial Surgery, University of Frankfurt am Main, Germany
3Division of Emergency Medicine, Katharinen hospital Stuttgart, Germany
4Division of Emergency Medicine, University of Ulm, Germany
5Division of Oral and Maxillofacial Surgery, Marienhospital Stuttgart, Germany
*Corresponding Author: Adrian Hoenle MD, BSc, Division of ENT/Head and Neck Surgery, Marienhospital Stuttgart, 70199 Stuttgart, Germany
Abstract
Objectives: Epistaxis is a common and potentially life-threatening medical condition with approximately 60 percent of the population experiencing it at least once in a lifetime. During the COVID-19 pandemic, there is cause for concern that patients with different kinds of medical symptoms are not seeking medical attention because of fear of being exposed to SARS-CoV-2. Therefore, we aimed to determine, whether there was a decline of emergency department visits with epistaxis after the pandemic-related lockdown.
Study Design: A retrospective analysis of all patients that were treated with epistaxis according to the ICD-10-code in the timespan from February 1st to June 8th, 2020, at Marienhospital Stuttgart, Germany.
Results: This retrospective review showed an overall decrease of patients during the lockdown period but also contrary results according to age groups. The percentage of older patients with epistaxis increased during the lockdown, whilst there was a decline of younger patients. However, none of these findings were strong enough to be statistically significant.
Conclusion: A decrease in younger patients with epistaxis during the lockdown period might be caused by fear of exposure to COVID-19. We further assume that epistaxis is a severe medical condition that is not underestimated even during this pandemic in the elderly.
Keywords: Covid-19; Epistaxis; Emergency Department; Lockdown; Pandemic
Introduction
Epistaxis
Epistaxis is a common ENT the emergency of varying severity as approximately 60 percent (%) of the population experience it at least once in a lifetime.[1] It can be caused by many different reasons ranging from idiopathic to even cancerous lesions.[2] 6 % of these patients require medical or surgical attention as most cases can be managed conservatively.[3] The most common cause is the idiopathic or spontaneous form of epistaxis which accounts for at least 70 % of cases and is often related to hypertension, atherosclerotic disease, smoking, or oral anticoagulation.[4] It has been shown that the incidence of epistaxis increases with age.[5]
Approximately 90 % of epistaxis arise from the little area along the anterior septum.[6] This area is supplied with blood by the Kiesselbach plexus which is composed of second-order branches of the external and internal carotid arteries. [7] Hemorrhage here is commonly being referred to as anterior epistaxis and can usually be managed by applying pressure to the nostrils, topical vasoconstricting or haemostatic agents, cryotherapy, electrocautery, or anterior nasal packing.[8]
Epistaxis originating from the more posterior parts of the nasal cavity are referred to as posterior epistaxis and accounts for only 5 % to 10 % of cases.[9] In these cases, the management of posteriorly based nasal bleeding with the application of anterior and posterior nasal packs are less successful, ranging from 48 % to 83 %.[10-12] In the remaining patients, nasal hemorrhage either continues despite nasal packing or reoccurs after removal of the nasal packs. For this reason, posterior epistaxis can be treated via an endoscopic or open surgical approach with direct ligation or cauterization of the involved artery.[13] Surgical treatment of epistaxis has been reported to show a success rate of 97 %.[14]
Endovascular embolization is an effective alternative to halting nasal bleeding with reported success rates of 71 % to 100% and usually minor complications like septal perforation, sinusitis, headache, facial or jaw pain as well as facial edema or otitis media.[2,13] However major complications resulting from inadvertent embolization can occur and include stroke, facial nerve paresis, soft-tissue necrosis, and even blindness.[14-16] These complications rarely arrive with ligation of the involved artery or nasal packing and are often caused by continued bleeding from the ethmoidal branches of the ophthalmic artery.[6,13]
Considering the latest research, epistaxis may be a presenting symptom of severe acute respiratory syndrome coronavirus-2 (SARS- CoV-2), as this virus impacts nasal epithelium and therefore possibly increasing the epistaxis risk.[17]
Emergency department visits during COVID-19
With iatrophobia being a major factor in delaying emergency treatment during the pandemic, there is cause for concern that patients with different kinds of medical symptoms are not seeking medical attention in the emergency department (ED).[18,19] A Canadian study showed a significant decrease of 35 % in the number of strokes referrals during a provincial lockdown and after the first confirmed COVID-19 cases were officially reported in the province.[20]
In the United States, ED visits for acute life-threatening health conditions such as myocardial infarction, hyperglycaemic crisis, and stroke decreased rapidly as shown in different studies.[21-26] In the 10 weeks after the emergency declaration, ED visits declined 23 % for myocardial infarction, 10 % for a hyperglycaemic crisis and 20 % for stroke compared with the preceding period. This crucial reduction in ED visits for life-threatening conditions during the COVID-19 pandemic might be explained by many pandemic-related factors such as fear of exposure to COVID-19, unintended consequences of public health recommendations to minimize non-urgent healthcare or stay- at-home orders.[27] Either way a short-term decline in the incidence of these conditions is unlikely.
Aim of the study
We aimed to determine whether there was a decline of ED visits with epistaxis after the pandemic-related closing of businesses and borders also referred to as lockdown. In our opinion, the experience of epistaxis is an obvious medical symptom, whereas other severe conditions such as stroke could be underestimated concerning possible exposure to COVID-19.[18, 28]
Methods
Study design and participants
This epidemiological study was executed retrospectively so that no direct patient contact has been made and was solely performed at Marienhospital Stuttgart (Stuttgart, Germany). Before being granted access to the emergency department, patients were audited for symptoms of COVID-19 like fever, cough, or anosmia.[29-31] Additionally, all our ENT doctors have been advised to ask for symptoms of COVID-19 as well before treating patients in the emergency department. This advice dates back to January 28th, which is the day after the first confirmed COVID-19 case in Germany. The application to perform this study was approved by the hospital’s ethics committee in October 2020.
The space of time for the case group lockdown was set from March 15th to April 26th concerning the closing of businesses and borders. The space of time for the control groups was set to be from February 1st to March 14th in the pre lockdown group and from April 27th to June 8th in the post lockdown group. This resulted in a total period of 18 weeks.
We included all patients that were treated with epistaxis according to the ICD-10-code in the timespan mentioned above and if the arrival at our emergency department was due to an acute and unplanned event. Non-emergency patients, for instance, patients with planned appointments such as check-ups or changes of dressing material were excluded, even if this had led to secondary hemorrhage. Lack of the ICD-10-code in the database of Marienhospital Stuttgart also led to elimination.
Aside from the diagnosis, the collected data included the patient’s sex and birthday. Starting from the patient’s birthday, the age was calculated in years.
Statistical analysis
Descriptive analyses were used to summarize baseline characteristics of the study participants. The data are presented as median as well as percentages (%) and absolute numbers (n). Differences were also calculated as percent and percentage points (% P).
The Mood’s median test was used to compare the medians of the various age and sex groups.[32] To compare the alterations between the three specified periods we used Pearson’s chi-squared test with the theoretical distribution being constant values for all time points.[33] To test for a linear trend in proportions we used the Cochran-Armitage test for trend.[34] The Bonferroni correction was used to counteract the problem of multiple comparisons.[35] A p-value of < 0,05 was considered to be statistically significant. Statistical analyses were performed using Prism, version 9.0.0 (GraphPad Software, San Diego, CA, USA) and Microsoft Excel, version 2019 (Microsoft Corporation, Redmond, WA, USA).
Results
In this study, we included a total of 151 patients between the age of 15 and 97 years of age (median: 72 years).
Fragmented into the three-time points we found medians of 65 years (pre lockdown), 78 years (lockdown), and 67 years (post lockdown) of age without statistically significant differences (n.s.). Concerning these differences, the median age increased by 20 % (13 years) during the lockdown and decreased by 15 % (12 years) afterward.
Cases according to gender
56% (85/151) of the included patients were male, 44 % (66/151) were female.
Table 1. The number of male patients according to the three periods and overall.
|
|
Male |
|||
|
|
number of patients (n) |
the fraction in the respective group (%) |
p-value |
p-value (trend) |
|
Pre-Lockdown |
32 |
60 |
n.s. |
n.s. |
|
Lockdown |
28 |
60 |
n.s. |
n.s. |
|
Post Lockdown |
25 |
49 |
n.s. |
n.s. |
|
Total |
85 |
56 |
n.s. |
n.s. |
The account of male patients decreased slightly by 1 3 % (4/85) between pre-lockdown and lockdown. In the post lockdown group, the number further decreased by 11 % (3/85).
Table 2. The number of female patients according to the three periods and overall.
|
|
Female |
|||
|
|
number of patients (n) |
the fraction in the respective group (%) |
p-value |
p-value (trend) |
|
Pre-Lockdown |
21 |
40 |
n.s. |
n.s. |
|
Lockdown |
19 |
40 |
n.s. |
n.s. |
|
Post Lockdown |
26 |
51 |
n.s. |
n.s. |
|
Total |
66 |
44 |
n.s. |
n.s. |
The account of female patients decreased slightly by 10 % (2/66) between pre-lockdown and lockdown. In the post lockdown group, the number increased by 37 % (7/66).
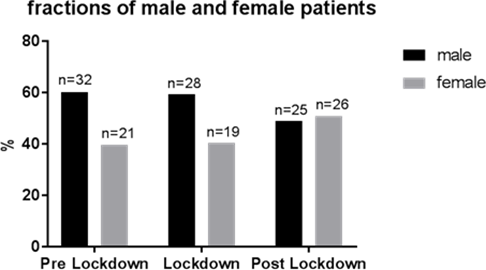
Figure 1. Fractions of male and female patients according to the three periods. Data are shown as percentages (%) and absolute numbers (n).
In total, we did not find any statistically significant differences neither concerning gender nor concerning the time points.
Cases according to age groups
The exact split between the five age groups as well as the median of each group (p < 0,0001) is shown in the table below.
In total, we did not find any statistically significant differences neither concerning gender nor concerning the time points.
Cases according to age groups
The exact split between the five age groups, as well as the median of each group (p < 0,0001), is shown in the table below.
Table 3. Number of patients according to age group and overall.
|
Age group (years) |
number of patients (n) |
Median (years) |
|
15-44 |
29 |
28 |
|
45-64 |
34 |
58 |
|
65-74 |
19 |
70 |
|
75-84 |
49 |
81 |
|
> 85 |
20 |
89 |
|
Total |
151 |
72 |
Table 4 and figure 2 show the mentioned age groups divided into the defined time points.
Table 4. Percentages of patients according to age group and period.
|
Age group (years) |
Pre-Lockdown (%) |
Lockdown (%) |
Post-Lockdown (%) |
p-value |
p-value (trend) |
|
15-44 |
31 |
17 |
52 |
n.s. |
n.s. |
|
45-64 |
50 |
24 |
26 |
n.s. |
n.s. |
|
65-74 |
37 |
26 |
37 |
n.s. |
n.s. |
|
75-84 |
26 |
41 |
33 |
n.s. |
n.s. |
|
> 85 |
35 |
45 |
20 |
n.s. |
n.s. |
|
Total |
36 |
30 |
34 |
n.s. |
n.s. |
It is shown that the overall percentage of patients dropped by 11 % (6 % P) during the lockdown and increased by 9 % (4 % P) during the post lockdown phase.
The age group of 15-44 years showed a decrease of 44% (14 %P) during the lockdown with an increase of 200 % (35 %P). The age group of 45-64 years showed a greater decrease of 53 % (26 %P) during the lockdown phase and a slight increase of 13% (2 %P) afterward.
In the group of 65-74 years, we also found a decrease in the lockdown group of 29 % (11 %P) with an increase of 40 % (11 %P) in the post lockdown phase.
In contrast to these results, we found an increase during the lockdown phase amongst the two eldest age groups. Between 75 and 84 years we found an increase of 54 % (15 %P) and 29 % (10 %P) for the age group greater than 85 years. In the subsequent post lockdown period, the cases dropped by 20 % (8 %P) in the 78-84 years age group respectively 56 % (25 %P) in the oldest age group. However, none of these differences were strong enough to be statistically significant nor did we find a significant trend of increasing or decreasing cases.
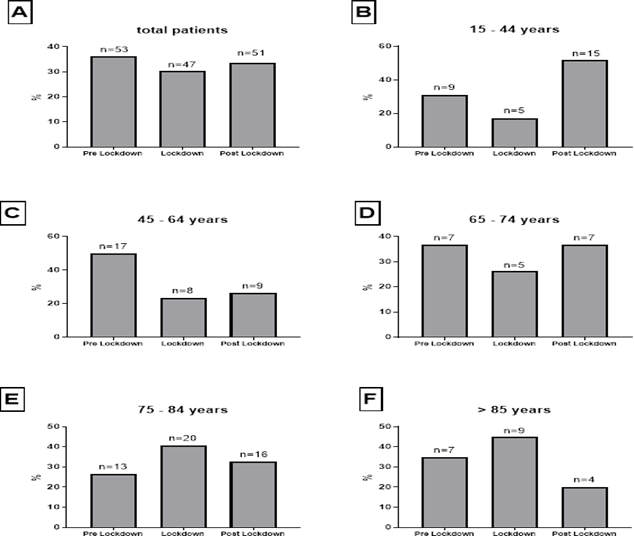
Figure 2. Fractions of patients according to age group and period. Data are shown as percentages (%) and absolute numbers (n).
Discussion
This study aimed to determine, whether there was a decline of ED visits with epistaxis during the lockdown with a time span of 6 weeks, compared to ED visits 6 weeks prior and 6 weeks after this period. In our opinion, the experience of epistaxis is an obvious medical symptom, whereas other severe conditions such as stroke could be underestimated concerning possible exposure to COVID-19.[18, 20, 27, 28] The decline in patients with different, severe medical symptoms during the lockdown has been reported in numerous studies.[21-26]
We showed that the median age increased during the lockdown period which might be caused by younger people who have stayed away from an ED visit as previous studies showed a higher possibility of an ED visit with increasing age.[36, 37] With the incidence of epistaxis increasing with age, it is not surprising that the majority of our patients are older.[5] More specifically our results show a decline of patients with epistaxis younger than 74 years of age during the pandemic-related lockdown. This might be caused by pandemic-related factors such as fear of exposure to COVID-19, unintended consequences of public health recommendations to minimize non-urgent healthcare or stay-at-home orders as mentioned above.[27] The reason for these findings not being statistically significant – and therefore possibly being coincidental – could as well be caused by the small number of patients in the respective groups. Additionally, our results show an increased number of patients during the lockdown period above the age of 75 years. The reason for this is unknown. Overall, our results show a decrease of patients during the lockdown period which could be caused by the reasons mentioned above even if the effect was not strong enough to be statistically significant.
In our opinion, the small changes between the pre-lockdown and lockdown period concerning the patient’s gender are extraneous, given the small number of patients overall.
Limitations
This study features some limitations. Firstly, no COVID-19 PCR tests have been made to rule out a COVID-19 infection for sure. Concerning epistaxis being a possibly life-threatening medical condition, we were restrained to a careful anamnesis. This ensured a rapid treatment initiation for the patients. Furthermore, we were unable to split up the included patients according to the different types of epistaxis or underlying medical causes as the total number would have had to be much higher.
Conclusion
Although not being statistically significant we found a decrease in younger patients during the lockdown period which might be caused by fear of exposure to COVID-19, unintended consequences of public health recommendations to minimize non-urgent healthcare, or stay-at-home orders. These findings match with results from previous studies.21-26 For an unknown reason we found an increase of patients aged 75 years and older. This corresponds with another study performed at our hospital, which is yet to publish. Therefore, it can be assumed that epistaxis is a severe medical condition that is not underestimated even concerning possible exposure to COVID-19 in the elderly.
Bullet Point Summary
-
Increased number of patients during the lockdown period above the age of 75 years
-
Decrease in younger patients during the lockdown period which might be caused by fear of exposure to COVID-19
-
Epistaxis is a severe medical condition that is not underestimated even concerning possible exposure to COVID-19 in the elderly
Acknowledgments
Competing Interests: The authors report no conflicts of interest.
Funding: No funding was utilized for this research.
References
-
Tan LK, Calhoun KH (1999) Epistaxis. Medical Clinics of North America. 83(1): 43-56.
-
Garcia S, Albaghdadi MS, Meraj PM, Schmidt C, Garberich R, et al. (2020) Reduction in ST-segment elevation cardiac catheterization laboratory activations in the United States during COVID-19 pandemic. Journal of the American College of Cardiology. 75(22): 2871-2872.
-
Lechien JR, Chiesa-Estomba CM, De Siati DR, Horoi M, Le Bon SD, et al. (2020) Olfactory and gustatory dysfunctions as a clinical presentation of mild-to-moderate forms of the coronavirus disease (COVID-19): a multicenter European study. European Archives of Oto-Rhino-Laryngology. 277(8): 2251-2261.
-
Pearson K. (1900) X. On the criterion that a given system of deviations from the probable in the case of a correlated system of variables is such that it can be reasonably supposed to have arisen from random sampling. The London, Edinburgh, and Dublin Philosophical Magazine and Journal of Science. 50(302): 157-75.
-
Miller RG (1981) Simultaneous statistical inference. NY: McGraw-Hill. 1967.



